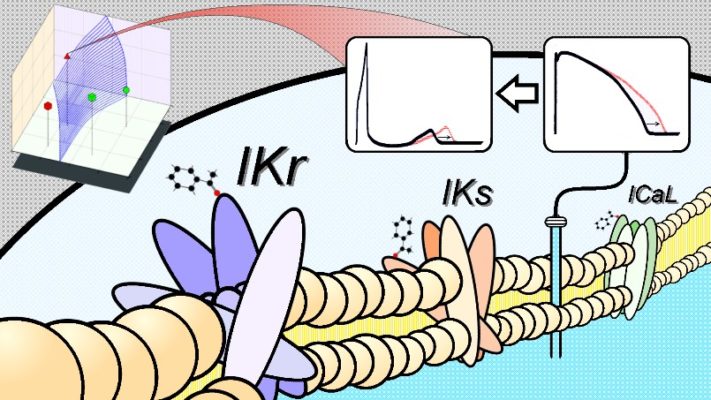
Computational simulation for personalised prediction of drug effects on the cardiac activity
Cardiac tachyarrhythmias are one of the main causes of morbidity and mortality in developed countries. In spite of intense scientific research, their treatment is still far from being satisfactory and anti-arrhythmic drugs may be inefficient and even pro-arrhythmic under certain circumstances, as shown by CAST trials. Moreover, some noncardiovascular drugs have led to potentially lethal arrhythmias. As efficacy and security of drugs depends on the patient and genetic defects play a relevant role, this proposal will address the development of bioinformatic models to allow the personalisation of the antiarrhythmic therapy.
The current approach to drug discovery and development, highly based on trial-and-error processes, is largely inefficient, extremely lengthy and risky for pharmaceutical industries and too much costly for society. Pharmaceutical companies have started to introduce computational simulations in this process, like QSAR modeling. However, application of computational modeling of the electrical activity of the heart to study of the effects of the drugs is more recent and it is barely used. Recently, an international initiative called Comprehensive in-vitro Pro-arrhythmia Assay (CiPA), led by the American Food and Drug Administration, is assessing the possibility of using the modelling and simulation of the cardiac electrophysiological activity to predict the effects of drugs on the proarrhythmic risk.
To improve the current pharmacological treatments, this proposal will use mathematical models of the electrical activity of the heart to assess the cardiotoxicity of drugs and the efficacy of antiarrhythmic drugs. Additionally, the most proarrhythmic kinetic properties of drugs and the ideal kinetic properties of anti-arrhythmic drugs will be revealed. To do this, the state-of-the-art of computational modelling will be moved a step further and a wide variety of dynamic drug-ion channel interactions will be systematically simulated. In addition, multi-scale computer-based models from the ionic to the organ level will be updated to simulate the effects of drugs on cardiac arrhythmias generation, maintenance and termination, such as ventricular tachycardia/fibrillation. These simulations will be extended, including patient´s sex, to individuals with channelopathies, such as the long QT syndrome or the catecholaminergic polymorphic ventricular tachycardia, whose pharmacological treatment could be personalized.
Generation and simulation of prototypical models of drug action in virtual patients is a promising, efficient, cost-effective and ground-breaking methodology that can reveal fundamental biological principles and mechanisms which can improve the design and development of drugs in the future. There is no reasonable, efficient and cost-effective alternative experimental or clinical strategy that can achieve it in a such effective and economic way. Besides its scientific and technical impact, this proposal has a high social and economic potential as its results will reveal new insights into the generation, maintenance and termination of cardiac arrhythmias and could be used to improve patients’ treatment outcomes and reduce costs in the design of drugs.
In progress…